What is a Molten Carbonate Fuel Cells?
Think of a battery. You use it, it dies, and you throw it away or recharge it. Now, imagine a battery that never dies as long as you keep feeding it fuel. That is a fuel cell.
A Molten Carbonate Fuel Cells (MCFC) is a special type of fuel cell. It is a machine that turns gas (like natural gas or hydrogen) directly into electricity. It does not burn the gas like a car engine. Instead, it uses a chemical reaction.
MCFCs are the “heavy lifters” of the fuel cell world. They are big, stationary, and run at extremely high temperatures.
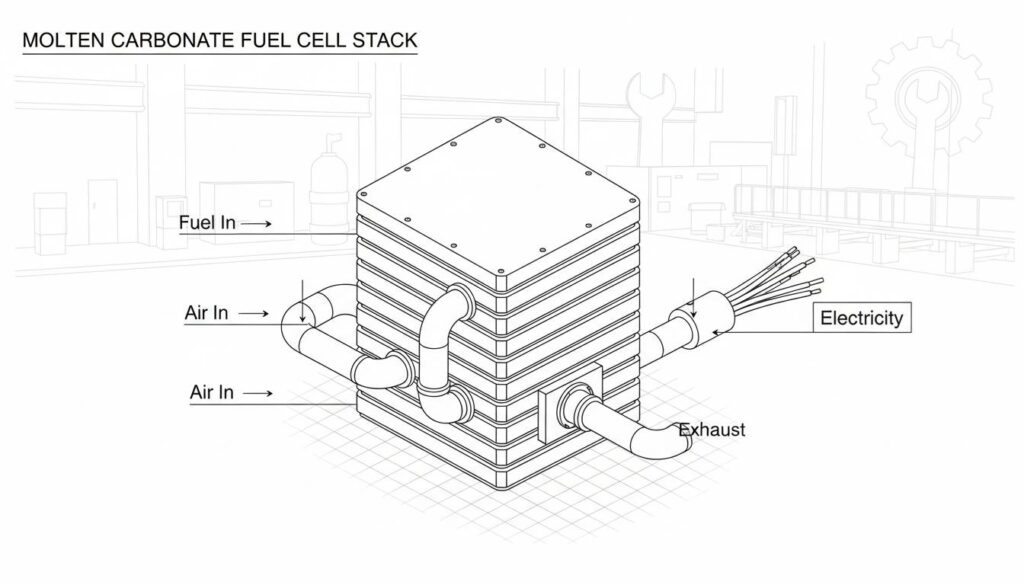
Technical Figure: A simplified 3D diagram of a Molten Carbonate Fuel Cell stack. It looks like a large, rectangular metal box with pipes entering and exiting. One pipe is labeled ‘Fuel In’, one is ‘Air In’, and wires are coming out labeled ‘Electricity’. The background is a clean industrial setting.
The “Molten” Part: The Secret Sauce
Why do we call it “Molten Carbonate”? It is all about the electrolyte.
Every battery or fuel cell has an electrolyte. This is the material inside that allows electricity-carrying particles (ions) to move from one side to the other.
In an MCFC, the electrolyte is a mixture of salts (carbonates).
- At room temperature: It is a solid, like table salt.
- At operating temperature (650°C / 1200°F): It melts. It becomes a hot, liquid soup.
This liquid salt is excellent at conducting ions.
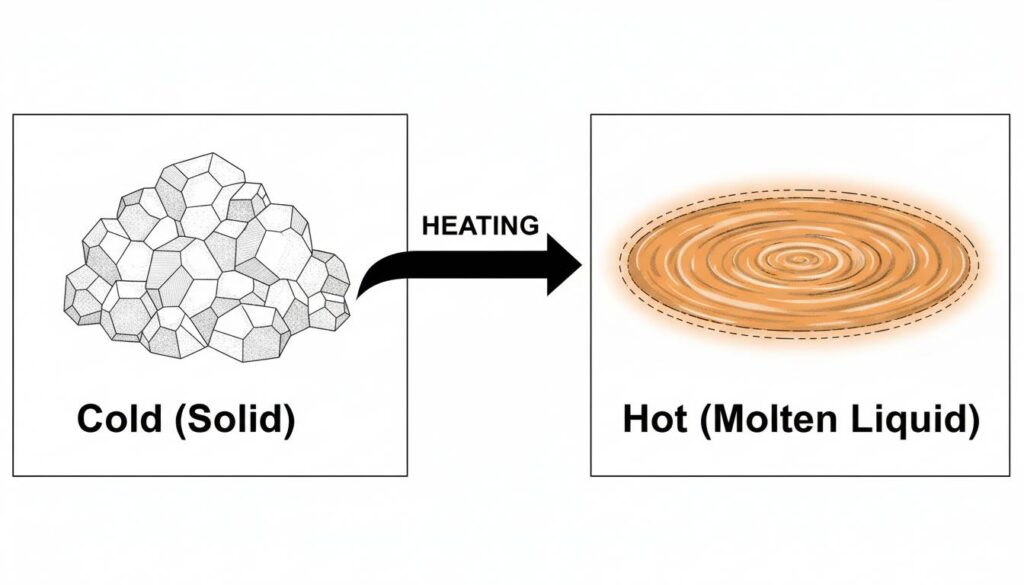
Technical Figure: A split-screen comparison illustration. On the left, a pile of white salt crystals labeled ‘Cold (Solid)’. On the right, a glowing orange liquid pool labeled ‘Hot (Molten Liquid)’. An arrow points from left to right indicating heating.
If the salt inside the fuel cell is solid at room temperature, what do you think happens if we turn the machine off and let it cool down completely? How might that make starting it up again difficult?
How Does It Work?
The MCFC works like a sandwich. It has three main layers.
- The Anode: The negative side (Fuel side).
- The Electrolyte: The hot, salty liquid in the middle.
- The Cathode: The positive side (Air side).
Step 1: The Cathode (The Air Side)
We pump air and Carbon Dioxide (CO2) into the Cathode side. Because the cell is so hot, the oxygen in the air reacts with electrons coming from the wire. They combine with CO2to form Carbonate ions.
Think of these ions as little boats carrying oxygen. They dive into the hot, liquid electrolyte “ocean.”
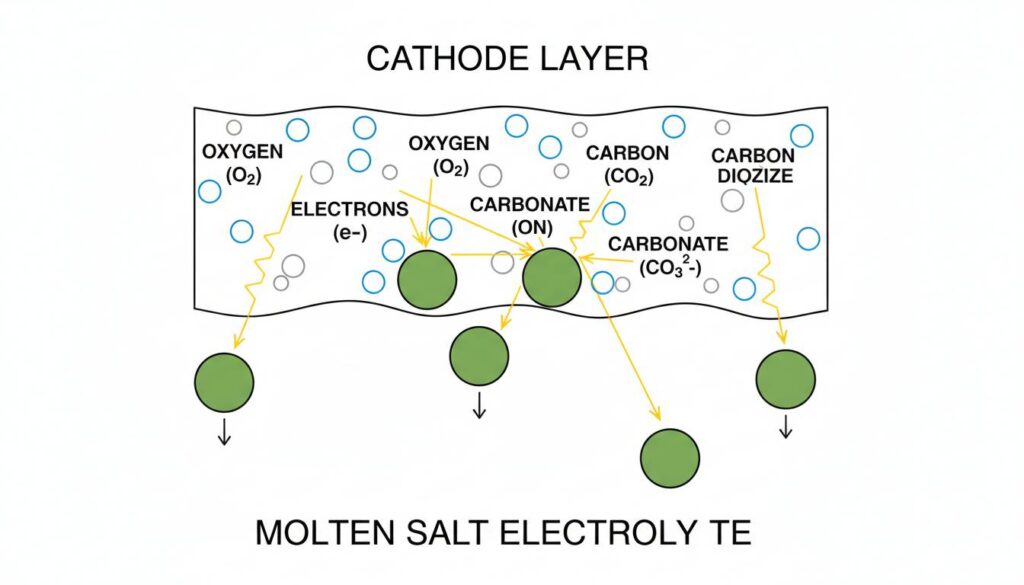
Technical Figure: A close-up cross-section of the Cathode layer. Blue spheres representing Oxygen and grey spheres representing Carbon Dioxide combine with small yellow sparks (electrons) to form larger green spheres (Carbonate ions). These green spheres are shown sinking into a liquid layer below.
Step 2: The Journey
The Carbonate ions swim through the molten salt. They move from the Cathode to the Anode. They are looking for fuel.
Step 3: The Anode (The Fuel Side)
On the other side, we pump in Hydrogen (the fuel). The Carbonate ions arrive and meet the Hydrogen.
A reaction happens immediately:
- The Carbonate ion gives its oxygen to the Hydrogen.
- This makes Water (H2O) and Carbon Dioxide (CO2).
- Most importantly, this reaction releases Electrons.
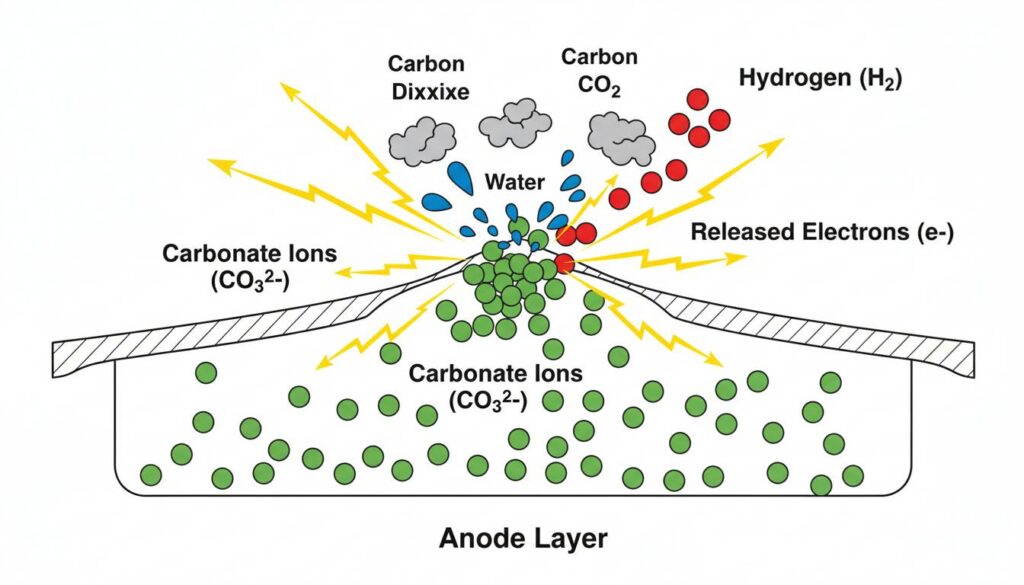
Technical Figure: A close-up cross-section of the Anode layer. Green spheres (Carbonate ions) rising from the liquid meet red spheres (Hydrogen). They collide and burst into blue water droplets, grey CO2 clouds, and bright yellow lightning bolts representing released electrons.
Step 4: The Electric Flow
The electrons are trapped. They cannot swim back through the salt. They have to find another way home.
We provide a wire connecting the Anode to the Cathode. The electrons rush through this wire. Moving electrons = Electricity. This powers our lights, computers, or factories.
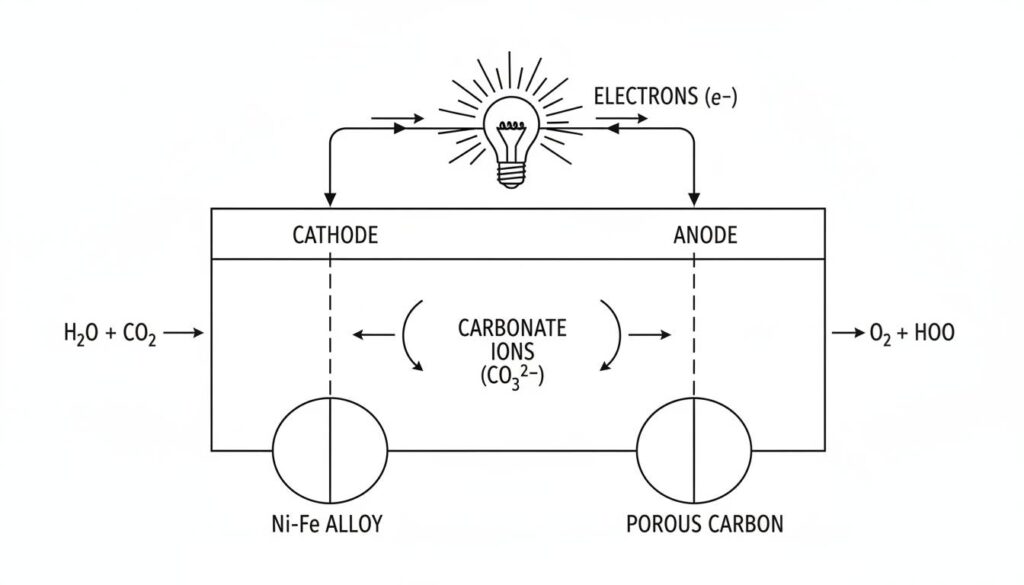
Technical Figure: A full schematic diagram showing the cycle. Arrows show Carbonate ions moving through the center liquid from right to left. Arrows show electrons moving through an external wire from left to right, passing through a lightbulb which is glowing.
In this process, we put Carbon Dioxide (CO2) into the Cathode, but the Anode also produces CO2. Can you think of a way to recycle the CO2 from the Anode back to the Cathode so we don’t waste it?
Why Are MCFCs Special?
MCFCs are different from the fuel cells used in space or in hydrogen cars.
High Efficiency (Cogeneration)
Because MCFCs run at 650°C, they are incredibly hot. Usually, heat is waste. But here, we can use it.
We can use the hot exhaust to boil water and make steam. That steam can turn a turbine to make more electricity. This is called Cogeneration (Combined Heat and Power).
- Standard Generator: Wastes a lot of energy.
- MCFC with Cogeneration: Uses up to 85% of the fuel’s energy. That is extremely high!
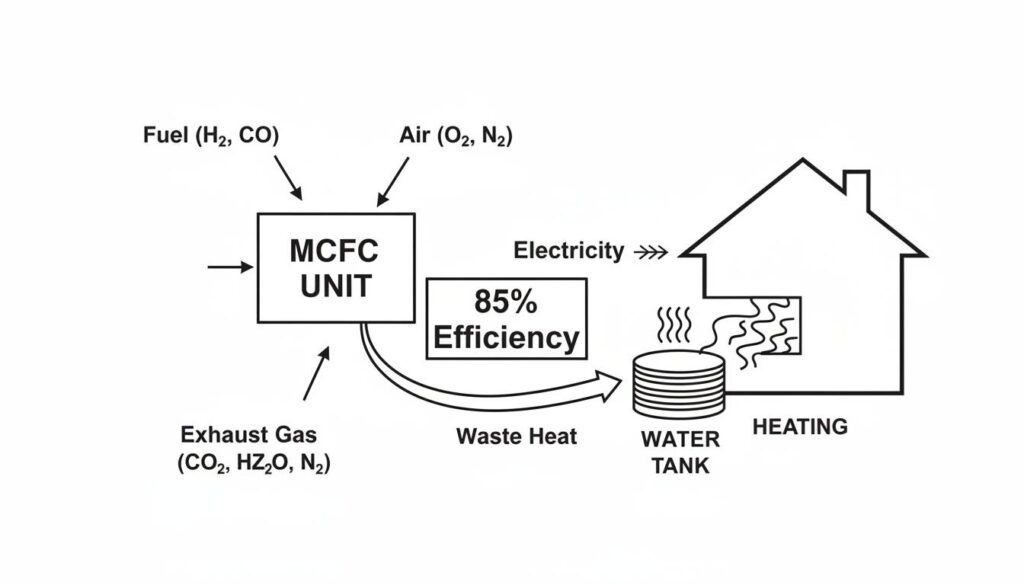
Technical Figure: An infographic showing an MCFC unit. An arrow labeled ‘Electricity’ points to a house. A second arrow labeled ‘Waste Heat’ points to a water tank, turning it into steam to heat the building. The text ‘85% Efficiency’ is highlighted.
No Expensive Jewelry Metals
Many fuel cells need Platinum to work. Platinum is a precious metal used in expensive jewelry. It acts as a catalyst (a helper) to start the reaction.
Because MCFCs are so hot, they don’t need a helper. The heat forces the reaction to happen. This means we can use cheaper metals like Nickel inside the cell. This lowers the cost.
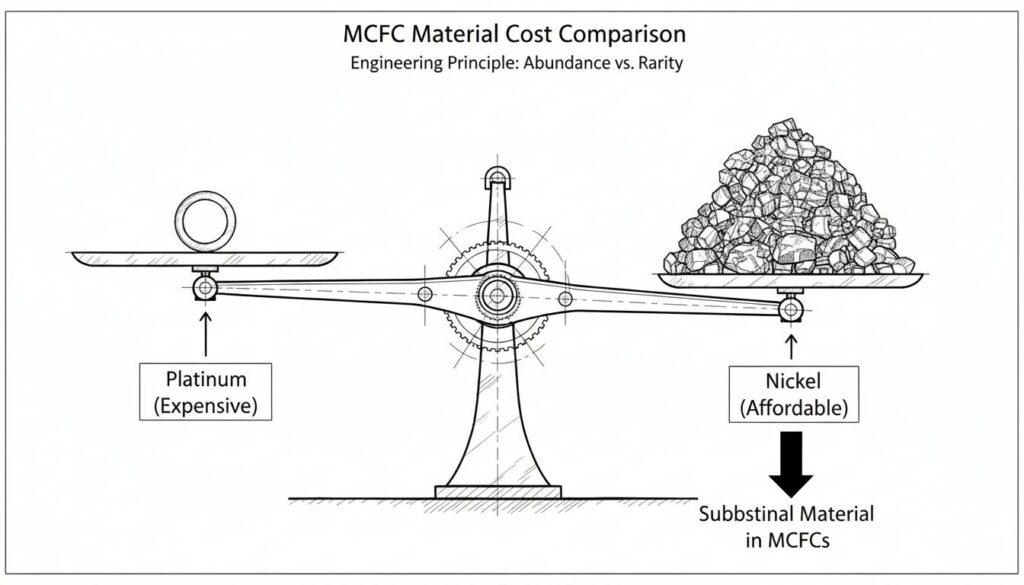
Technical Figure: A balance scale illustration. On one side, a small shiny ring labeled ‘Platinum (Expensive)’. On the other side, a large pile of industrial metal labeled ‘Nickel (Affordable)’. The Nickel side is heavier, showing it is the substantial material used in MCFCs.
Since MCFCs run so hot and don’t need expensive platinum, why don’t we use them in our cars or smartphones? Think about the temperature and the liquid salt.
The Downsides
Nothing is perfect. MCFCs have two main problems.
1. Corrosion
The electrolyte is hot, liquid salt. Salt water is very corrosive (it rusts metal). Hot liquid salt is even worse. It eats away at the metal parts of the fuel cell over time. This means MCFCs do not last as long as other types.
2. Slow Start-Up
You cannot just flip a switch and have power instantly. You have to melt the salt first. Bringing the cell up to 650°C takes a long time. Once it is on, you usually leave it on.
Summary
- MCFCs are high-temperature batteries that run on gas.
- They use molten salt as the electrolyte.
- They run at 650°C, which allows them to use cheap metals instead of platinum.
- They produce electricity and heat, making them very efficient for large buildings and factories.
