What is Saturated Vapor Pressure?
To understand hydraulic systems, we first need to understand liquids. You probably think water only boils when you put it on a stove. That is not true. Water (and oil) can boil even when it is cold.
Saturated Vapor Pressure is a specific pressure limit. If the pressure of a liquid drops below this limit, the liquid turns into a gas. It creates bubbles.
Think of a crowded elevator. The people are like liquid molecules packed tight. If the doors open and there is a giant empty space outside, people rush out and spread apart. They become like a gas. Vapor pressure is the force keeping them inside the elevator.
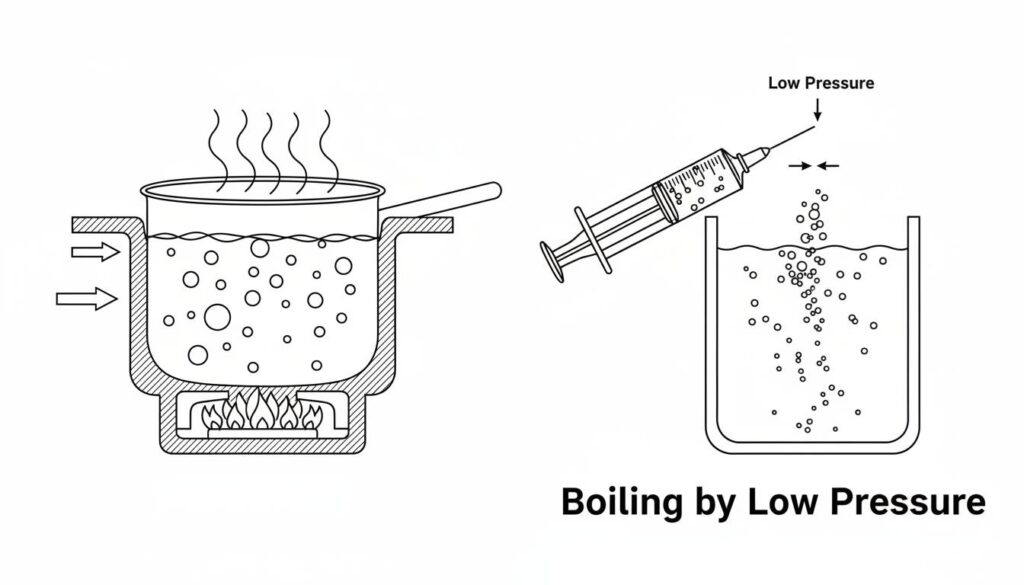
Technical Figure: A split-screen illustration. Left side: A pot of water boiling on a stove with fire underneath. Right side: A syringe filled with water being pulled back, creating a vacuum, showing bubbles forming in cold water without fire. Label the right side ‘Boiling by Low Pressure’.
The Soda Bottle Analogy
Imagine a sealed bottle of soda. You don’t see any bubbles. The pressure inside is high. This keeps the gas trapped in the liquid.
When you twist the cap open, you hear a hiss. The pressure drops instantly. Suddenly, bubbles appear everywhere. The liquid is trying to turn into a gas because the pressure went down.
In a hydraulic machine, we use oil instead of soda. But the rule is the same. If the pressure drops too low, the oil creates gas bubbles.
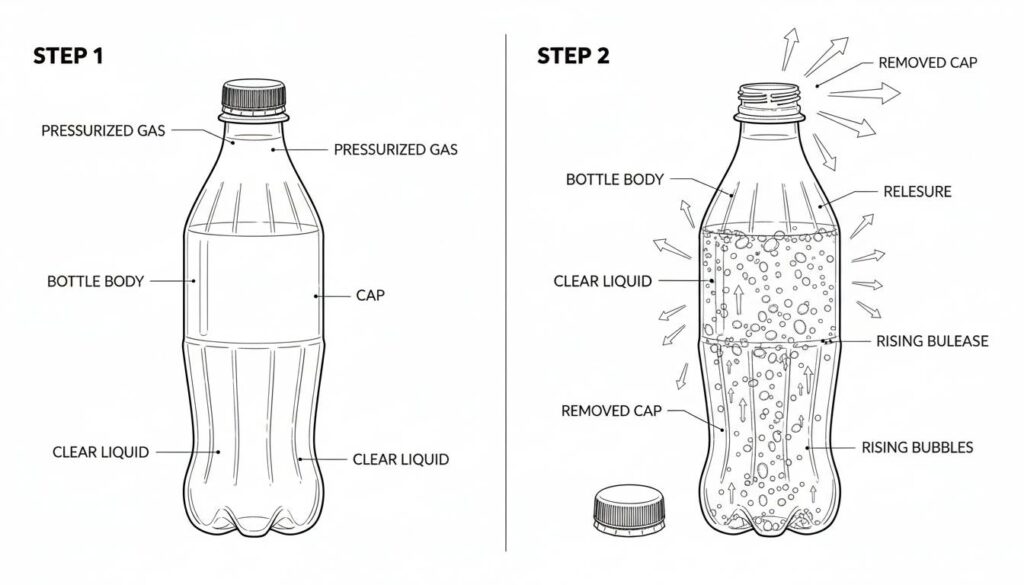
Technical Figure: A close-up diagram of a soda bottle. Step 1: Cap closed, liquid is clear. Step 2: Cap removed, pressure arrows pointing out, and many bubbles rising rapidly in the liquid.
Think About It:
If you went to the top of a very high mountain where the air pressure is low, would it be easier or harder to boil a pot of water? Why?
The Big Problem: Cavitation
Hydraulic systems use pumps to push oil. This oil moves heavy things, like the arm of a bulldozer. The pump sucks oil in from a tank. This “sucking” action lowers the pressure.
If the pump sucks too hard, the pressure drops below the saturated vapor pressure.
What Happens Inside the Pump?
- Bubbles Form: The oil “boils” instantly. It creates vapor bubbles.
- Bubbles Move: The pump pushes these bubbles into a high-pressure zone.
- Bubbles Collapse: The high pressure crushes the bubbles. They implode (explode inwards).
We call this process Cavitation.
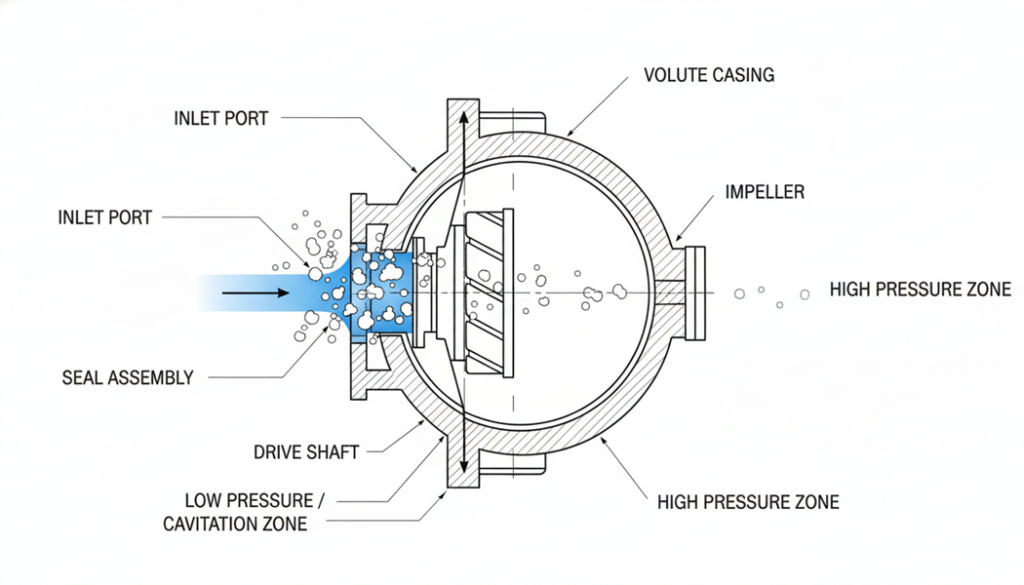
Technical Figure: A cross-section view of a hydraulic pump. Show blue liquid entering the inlet. Show white bubbles forming near the inlet where pressure is low. Show the bubbles disappearing/collapsing as they move to the outlet side.
Why Bubbles are Bad for Power
Hydraulic systems work because liquids are incompressible. This means you cannot squeeze them into a smaller space. It is like a solid rod of liquid.
Gas bubbles are compressible. You can squeeze them.
If your hydraulic oil has bubbles in it, the system feels “spongy.” When you pull the lever to lift a heavy load, nothing happens immediately. The pump is wasting energy squeezing the bubbles instead of moving the piston.
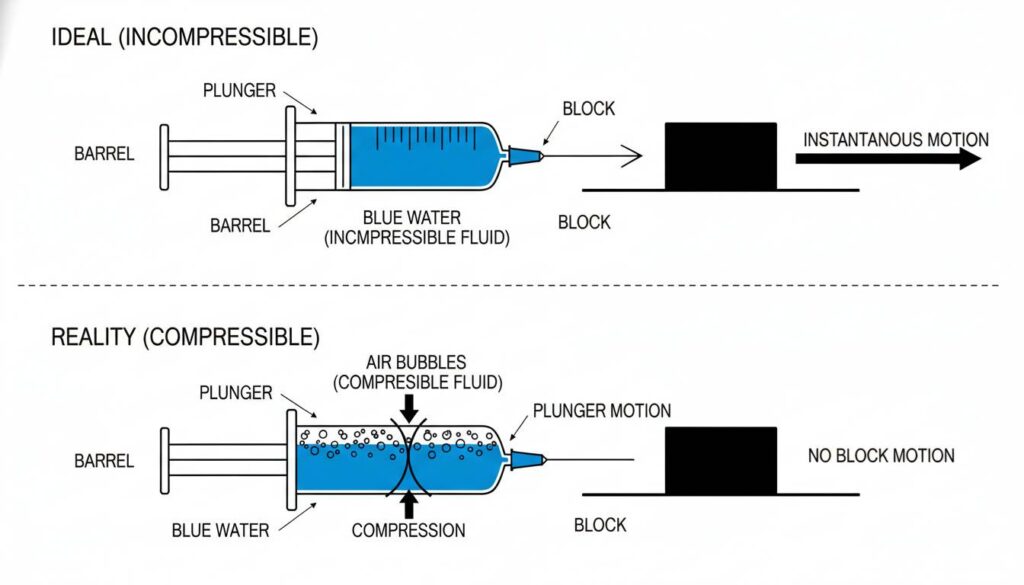
Technical Figure: A comparison diagram. Top: A syringe filled with blue water pushing a block; the block moves instantly. Bottom: A syringe filled with half water and half air bubbles; the plunger moves in, compressing the bubbles, but the block does not move.
Check Your Understanding:
Imagine your bicycle brakes are hydraulic (they use fluid). If air bubbles get into the brake line, what happens when you squeeze the brake handle? Will the bike stop instantly?
The Damage: Eating Away the Metal
Cavitation is not just annoying; it is destructive. When those vapor bubbles collapse, they don’t just pop gently. They snap shut with extreme force.
The Micro-Jet Effect
When a bubble collapses near a metal wall (like the inside of a pump), it shoots a tiny jet of liquid. This jet hits the metal like a bullet.
- Speed: The liquid jet moves incredibly fast.
- Heat: The collapse creates a tiny flash of extreme heat.
- Result: It chips away a piece of the metal.
Over time, this makes the metal look like it has been eaten by termites. This is called pitting. If this continues, the pump will break completely.
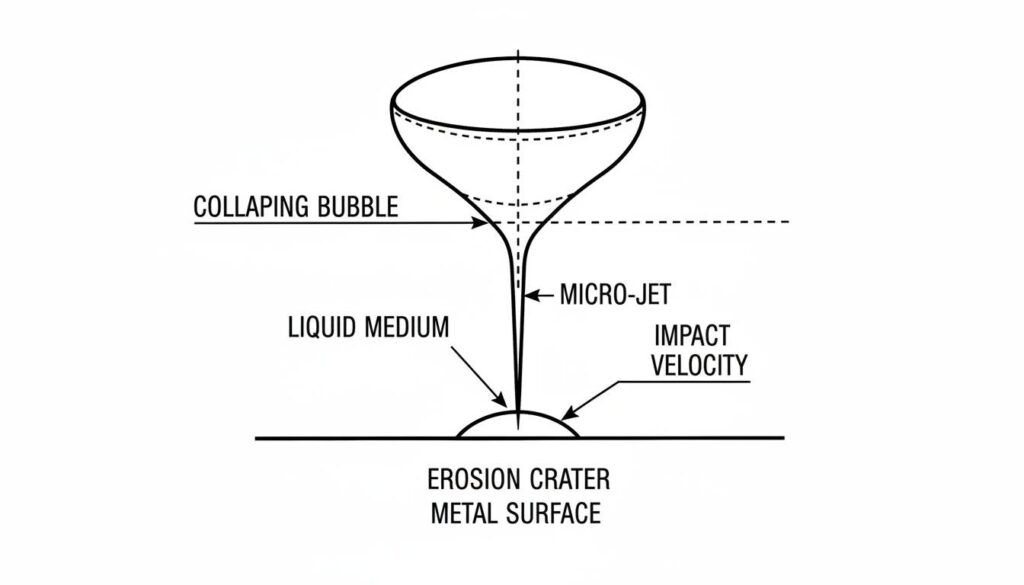
Technical Figure: A close-up technical illustration of a bubble collapsing against a metal surface. Show a ‘micro-jet’ of water piercing through the center of the bubble and hitting the metal wall, creating a small crater or pit.
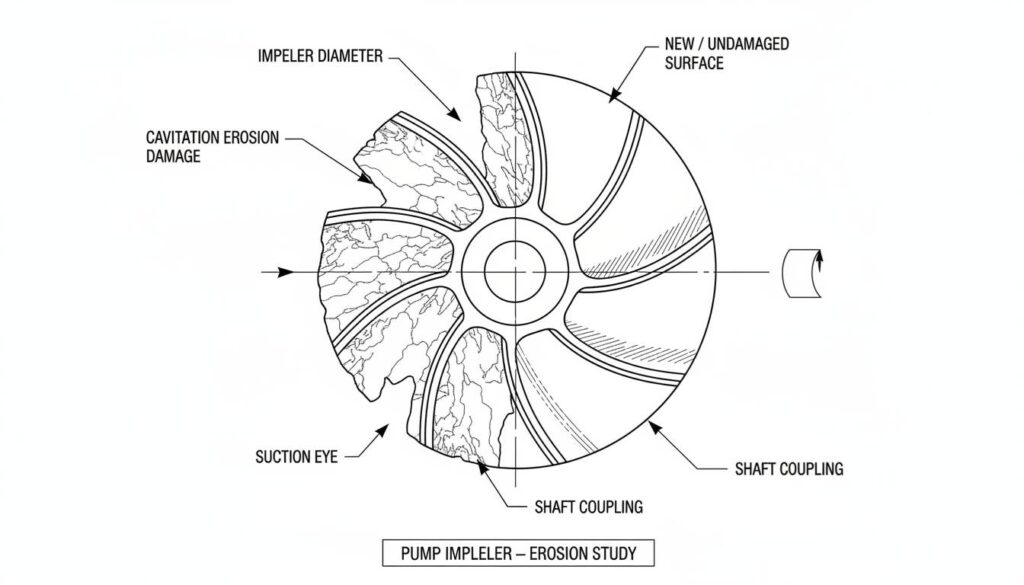
Technical Figure: A photo-realistic rendering of a damaged metal pump impeller. The surface should look rough, pitted, and eaten away, contrasting with a smooth, new metal surface.
How Engineers Fix This
To stop the oil from boiling (cavitation), engineers must keep the pressure above the saturated vapor pressure.
1. Keep the Inlet Pressure High
Engineers place the oil tank above the pump. Gravity helps push the oil into the pump. This keeps the pressure high enough so bubbles cannot form.
2. Keep the Temperature Low
Hot liquid boils easier than cold liquid. If the hydraulic oil gets too hot, its vapor pressure rises. This means it will bubble very easily. Engineers use oil coolers (like a radiator in a car) to keep the fluid cool.
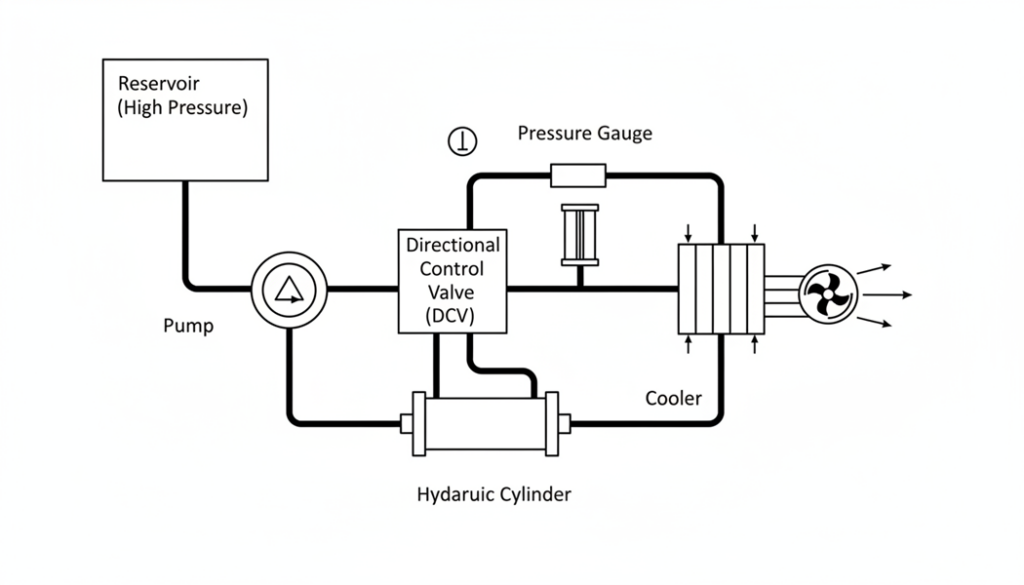
Technical Figure: A diagram of a hydraulic system layout. Show a reservoir tank located physically higher than the pump. Draw a ‘Cooler’ with a fan blowing on the pipes to keep the oil temperature down.
Final Thought:
If a hydraulic machine starts making a loud rattling noise like marbles shaking in a can, what do you think is happening inside the pump? Is the pressure too high or too low?
